MODAPLEX POLE/POLD1 Mutation
Analysis Kit
For improved decision-making:
A comprehensive POLE/POLD1 mutation panel
FEATURES
- Analyze 19 POLE and POLD1 mutations required for the risk classification of endometrial cancer
- Low DNA input – 4 ng per sample
- Benefit from an efficient workflow using a kit-specific data analysis software requiring no additional bioinformatics
- Verified with colorectal and endometrial cancer samples
Detection and differentiation of 19 somatic and rare germline mutations in the polymerase epsilon and polymerase delta-1 exonuclease domains is effectively achieved using the MODAPLEX POLE/POLD1 Mutation Analysis Kit. The assay enables researchers to advance promptly in clinical research.
SPECIFICATIONS
| Cat. No. | 85-10101-0050 | |
| Panel |
POLE EDMs: T278M, P286L, P286H, P286R, S297A, S297F, F367S, V411L(G>C), V411L (G>T), H422N, L424V, P436R, M444K, A456P, S459F, A465V POLD1 EDMs: D316N, C319Y, S478N |
|
| Reactions | 1 Multiplex-PCR per sample | |
| Kit content |
All reagents required to perform PCR are part of the MODAPLEX POLE/POLD1 Mutation Analysis Kit • POLE/POLD1 Primer Mix |
|
| Internal controls | 2 (POLE and POLD1 gene) | |
| PCR controls | 2 included (PC, NTC) | |
| Sample input | ~ 4 ng gDNA (FFPE) | |
| Sensitivity | Down to 2 % | |
| Turn-around time | ~ 4 h after nucleic acid preparation (including data analysis) | |
| Detection | Qualitative | |
| To be used with | MODAPLEX instrument | |
| Data analysis | MODAPLEX Reporter software |
Workflow
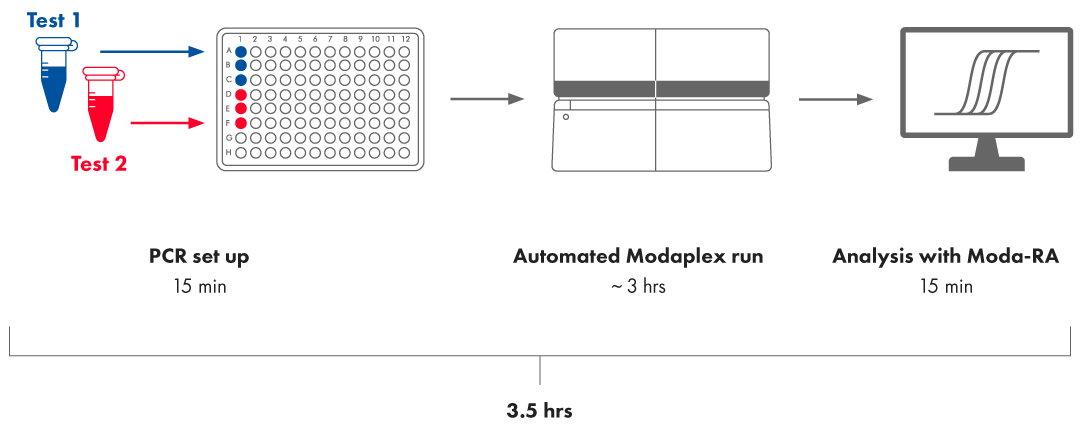
The Modaplex POLE/POLD1 Mutation Analysis Assays is to be used with the Modaplex instrument platform version 1.0 or higher. Thus, the POLE/POLD1 workflow is similar to the workflow of other Biotype Modaplex assays such as MSI Analysis Assay and comprises the same three steps after nucleic acid preparation: PCR set up, Modaplex run, and result interpretation.
Downloads
Order Information
| Product: | MODAPLEX POLE/POLD1 Mutation Analysis Kit |
| Size | 50 reactions |
| Cat. No. | 85-10101-0050 |
| Status | RUO* |
*RUO – Research Use Only products must be validated by the customer with clinically relevant material for diagnostic purposes.
To order the MODAPLEX POLE/POLD1 Mutation Analysis Kit, please e-mail us at sales@biotype.de